Climate change is a hot topic. Despite the experts telling us that ‘the
science is settled’ it just does not appear to be the case. This is the first in a series of Guides which describe the science issues in language that ordinary people can understand.
How did it all it start?
It really started not long after the Second World War when Rachel Carson wrote a book called The Silent Spring. She used that title to tell us that one year, when spring arrives, there may be no birds singing because they have all been killed by pollution. People started to realize that the environment was very important and that Mankind could destroy it or stop it from working properly.
A wonderful new insecticide had been discovered during the War called DDT. It was very cheap to make and very effective. It was soon being used everywhere to kill malaria-carrying mosquitos and insects that ate farm crops. Then scientists began to notice side effects. Some wild birds had stopped breeding properly because small amounts of left-over chemicals from DDT in the food-chain made their eggshells too thin and the eggs broke before their chicks had hatched properly. DDT chemicals were even found in birds in Antarctica tens of thousands of miles away from where it had been used.
At the same time people were also worried about nuclear weapons and the threat of nuclear war. For the first time the idea of wiping out all humans or all life on Earth appeared to be a real possibility.
These things really scared the hell out of people.
As a result the Environmental Movement sprang up. It is still going
today. Many of these problems have been fixed but people still feel very anxious about pollution and keeping our planet safe. It’s the only one we’ve got.
Why is Carbon Dioxide so important?
Back in the 1950’s Charles Keeling started making very precise and regular measurements of the amount of various gases in the atmosphere to see if they were changing. He did his experiments on a mountain in Hawaii a long way away from industry and traffic because he wanted to get an idea of what was happening on a global scale. These measurements are still being made in Hawaii today and in a number of other places such as the Base Line Monitoring Station at Cape Grim in Tasmania. All the measurements showed that one gas, Carbon Dioxide, CO2 , has been steadily increasing all over the world. At the same time the average temperature of the earth has also been increasing (but not quite so steadily).
Because the environment scare and the nuclear war scare were fresh in people’s minds they decided that the two things were connected and that rising CO2 must be causing the temperature to go up. They started calling CO2 a ‘pollutant’ like DDT and radioactive fallout. The increase in CO2 is supposed to be due to humans burning coal and oil in industry but there are other explanations for it.
Many scientists believe there has been an hysterical over-reaction to these observations and that, apart from the fact that both CO2 and temperature have both been increasing recently, there is really no evidence to connect the two things. It is just a delayed reaction to the ‘Future Shock’ of the scary 1950s.
But isn’t CO2 a greenhouse gas?
By ‘greenhouse gas’ we mean a gas that absorbs long-wave infrared radiation (LWIR). There are three main ones: CO2 , H2 O and O3 (ozone).
There is a simple mechanism by which greenhouses and cars get hot in the sun. Visible light from the sun comes in through the windows and heats the car seats. When things are heated they give off radiation with wavelengths that depend on the temperature. The sun is very hot so it gives out visible light and short-wave infrared. The car seats, although they are hot, are still much cooler than the sun so they give out LWIR. You can’t see it but you can feel it on your skin. The important thing is that LWIR doesn’t go through glass! So it gets trapped inside the car and the car gets hot.
In the same way sunlight comes in through the atmosphere and heats
the rocks and the grass and the sea which give off the LWIR which cannot get back out through the atmosphere because of the greenhouse gases so the earth stays warm just like the inside of a car in the sun.
If there were no greenhouse gases the earth would be −18 C(0 F) on average. This happens sometimes and we get an ice-age. Ice-ages are like leaving the car windows down.
Now here is the crunch: cars with thick glass windows don’t get any
hotter in the sun than cars with thin glass windows. Thin and thick glass both stop the LWIR almost completely.
Unless we are in an ice-age, adding more CO2 to the atmosphere
does not make it any warmer!
Then how does the earth lose the heat from the sun if the LWIR cannot get past the atmosphere? The earth should be getting hotter and hotter like a car in the hot sun.
I was only talking about radiation. There is another way that heat can get out. It is called convection: ‘hot air rises’. The most spectacular convection occurs during tropical storms. Hot moist air from the surface of the sea starts to rise. As it rises it expands and cools. As it cools water comes out as rain. This warms the air again so it rises even faster. Huge amounts of energy are released and the net effect is to carry heat from the surface of the ocean to the top of the atmosphere where the greenhouse gases are so thin that radiation kicks in. The excess heat is then radiated into space.
This is why the temperature of the tropical ocean seldom rises above
28 C (83 F). When it gets too hot, a tropical cyclone happens which cools everything down again.
Something similar happens at cooler latitudes but it is a lot less dramatic.
How do we know this? Everyone knows that as you get higher in the
atmosphere it gets cooler as you get away from the source of the heat.
True. There is snow at the top of mountains, even in the tropics.
If you compress a gas it gets hotter as anyone who has pumped up a bike tyre will know. We can predict how much hotter using the science of thermodynamics. If you let air expand it will cool down.
Imagine a parcel of air moving upwards. The pressure gets less and it
expands. When it expands it cools down. Thermodynamics tells us exactly how much it will cool down and exactly what the decrease in temperature will be for every meter you go up. For a stable atmosphere this is called ‘the adiabatic lapse rate’. It is measured hundreds of times a day with weather balloons.
Theory fits observation very well indeed. It has nothing to do with
greenhouse gases.
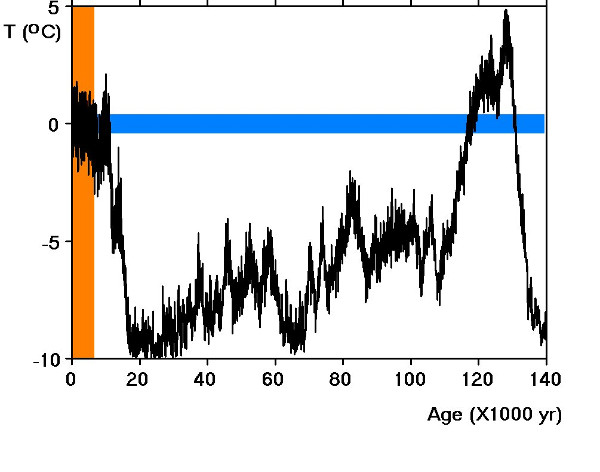
When CO2 and water vapour in the atmosphere increased at
the end of the last Ice Age, which had lasted for more than 80,000
years, it made the earth warm again . It made the big ice caps melt
and raised the level of the ocean. That happened 11,000 years ago
and created a boom time for Homo Sapiens (us). Apart from a few
random fluctuations, our climate has been remarkably warm and
stable ever since.
A pdf version of this post can be downloaded here: YPGTGE

